WRITTEN BY
Neil H Riordan, Luis Gerardo Jiménez Arias and Ramón Coronado
Submitted: September 9th, 2022 Reviewed: October 11th, 2022 Published: October 31st, 2022
DOI: 10.5772/intechopen.108541
Abstract
Numerous and diverse participants are involved in the development of novel therapies: patients, physicians, scientists, sponsors, governing bodies, lawmakers, institutional review boards, and bioethics proponents. While the welfare of the patient must always and unquestionably be at the forefront of any intervention along with informed consent, their wishes, their requests, and their expectations should also be considered at every step. The availability of stem cell research in various countries with dissimilar regulatory agencies has opened the door for thought-provoking questions about their validity from an ethical, legal, and moral perspective, which will be addressed in this chapter, framed within the doctor-patient relationship.
1. Introduction
In 2010, the Ministry of Health of Costa Rica disallowed stem cell therapy, citing concerns about the experimental nature of these procedures. This abrupt decision left several patients unable to continue receiving treatment at the San José hospital – most notably, a young pilot suffering from spinal cord injury after a 2008 airplane accident that rendered him paraplegic with no perspective of ever regaining mobility, according to two independent physicians. Having experienced encouraging progress in muscle recovery and bowel and sexual function following stem cell therapy, [1] he promptly filed a legal remedy for protecting his constitutional rights to the Supreme Court of Costa Rica, along with other patients in a similar predicament. In their appeal, they argued that patients had “a right to exhaust all technically feasible procedures to recover [their] health and quality of life”, citing Article XI of the 1948 American Declaration of the Rights and Duties of Man (the right to the preservation of health and well-being) [2]. This right should not, they contended, be limited by a political authority. Additionally, they invoked the American Convention on Human Rights, namely Articles 5 (the right to physical integrity) and 2 (whereby States Parties undertake to adopt (…) legislative or other measures to grant the rights or freedoms enshrined in The Convention). Lastly, none of the plaintiffs declared having experienced any adverse effects from their therapy at the time of the appeal; they had, in fact, perceived improvements in their health condition.
The Supreme Court ruled that medical treatments had to be permitted by law before their implementation, as had been argued by the defendant (the Ministry of Health). While the judges acknowledged the principle of patient autonomy, the doctor/patient relationship, human dignity, and the right to health, selecting a treatment should rely on the law. However, the Court also recognized that no adverse events had been noted throughout these treatment cycles with stem cells and that they had no factual or legal arguments to halt the treatments that had already begun. The plaintiffs were therefore permitted to continue their therapy.
Six years later, presidential decree n°39,986-S authorized regenerative therapies with adult stem cells in Costa Rica, based on national scientific recommendations, under the human right to health access principles, and citing decades of past research on the safety of hematopoietic stem cells transplants [3]. Therein are outlined the requirements to be submitted by those seeking stem cell therapy administration: safety profile, scientific rationale, cellular characterization, administration, and requirements for the qualifications of health professionals and facilities. While the decree makes a distinction between minimally manipulated and more than minimally manipulated cells, both are permitted with different avenues for authorization.
This Costa Rican case is pivotal because the Supreme Court ruling marks the first legal precedent in a Latin American country that allows patients who are already being treated with stem cells to continue their treatment – a first step, perhaps, to implement international legislation on stem cells. Following the law to the letter in that instance proved to be rather impractical, falling almost into irrationality as it limited the right of access to health for patients who required treatment as a last resort, and going against international law, which stipulated that Costa Rica must have taken the necessary legislative, economic and political measures to facilitate access to the said right to health. The subsequent presidential decree is also a landmark case since it authorizes using a non-minimally manipulated therapeutical product currently not permitted in other jurisdictions, notably in the USA according to the Federal Drugs Administration (FDA) guidance. The compassionate use of or access to drugs and new therapies remains limited on the grounds of minimizing harm to terminally ill patients [4]. How much evidence is necessary to release a given drug or a given experimental therapy? Are Phase I studies sufficient? Can effectiveness be shown at this stage? Answers remain unclear [4].
This chapter will examine legal and moral issues arising from stem cell treatments, the patient/doctor relationship, and the right of access to health and patient welfare in the context of current international regulations and medical tourism – and how conflicting regulations between countries pose conflicting views on ethics regarding patient access to new therapeutics.
2. Stem cells: definitions, applications, and considerations
Human “stem cells” is a broad term that may refer to various types of cells differing in their origin, applications, and characteristics. Hematopoietic stem cells, multipotent, self-renewing, and capable of generating blood cells, were first described in the early 60s [5]. Soon after, another type of multipotent stem cells differing from those of hematopoietic lineages were observed in bone marrow [6] – these would later become known as mesenchymal stem cells [7] or mesenchymal stromal cells (MSCs), with a limited capacity to differentiate into specific types of adult cells. Pluripotent embryonic stem cells (ESCs), capable of giving rise to all cells in the body, were derived in the late 90s from blastocysts [8]. Finally, induced pluripotent stem cells (iPSCs) were introduced in 2006: somatic cells reprogrammed to have the embryonic capacity for differentiation [9]. While much of the research in the 20th century was focused on isolating and identifying techniques to culture stem cells, the early 21st century saw a growing number of case reports and clinical trials seeking to establish their therapeutic potential. This line of research was not without challenges or controversy (Table 1).
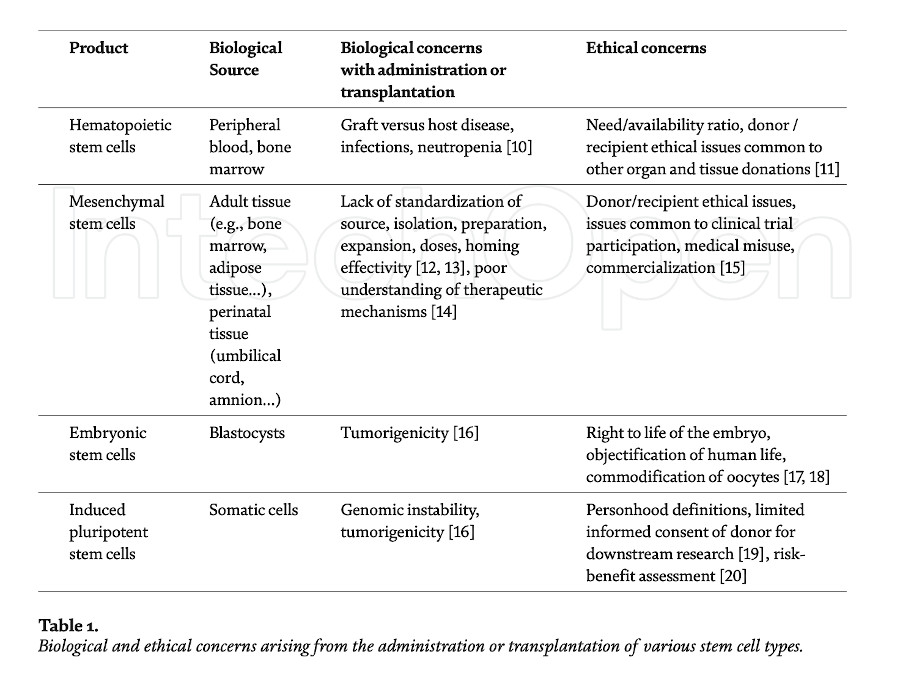
Most of the earlier bioethical controversy was focused, understandably, on the use of embryonic stem cells due to the loss of viable embryos in the process of isolation and derivation of the stem cells, which led to bans or limitations imposed on research in several nations. Mesenchymal stem cells, in contrast, share little of those ethical concerns related to their tissue origin since they can be derived from adult tissue, notably for autologous use, or from perinatal tissue, such as the umbilical cord that would routinely be discarded after a normal birth, for allogeneic use. Consequently, the ethical controversies surrounding the use of mesenchymal stem cells have shifted to considerations applicable to clinical trials and the development and commercialization of novel experimental therapies.
Recent clinical trials with mesenchymal stem cells cover a broad range of tissue sources and administration routes, complicating comparisons between published results. In brief, mesenchymal stem cells appear safe for administration at least in the short term, [21, 22, 23], and some long-term reports are also available [24, 25]. Adverse events are reportedly transient, usually fever or fatigue, and no tumorigenicity or malignancy has been observed thus far [23]. While the mechanisms of action of mesenchymal stem cells are not entirely understood, current research views stem cell secretions as key for their immuno-modulatory, anti-inflammatory, and therapeutic properties. The conditions treated in various clinical trials or case reports include cardiovascular, neurological, autoimmune, orthopedic, pulmonary, and graft-versus-host disease, [26] and more recently, COVID-19 [27]. At least ten products derived from mesenchymal stem cells have received regulatory approval in South Korea, Japan, India, Canada, Australia, and Europe [12]. The wide variety of conditions researched as well as the legitimacy of some products, may give the erroneous impression that mesenchymal stem cells are a “cure-all”: more research is necessary to standardize isolation and culture techniques, as well as to establish risk-benefits and true efficacy for certain conditions to avoid these pitfalls. Most importantly, trials should be conducted ethically, ensuring that disappointing results are also published, that all adverse events are carefully documented and reported, and that patients duly consent with a proper understanding of the procedures and the potential benefits and limits.
Throughout this chapter, “stem cells” should be understood to mean mesenchymal stem cells or adult stem cells unless otherwise indicated.
3. International regulation and medical tourism
The use of stem cells in Costa Rica has been commercially permitted since 2016. The current law does not specify the effectiveness of a treatment or the amounts to be charged, if any. The broad wording was designed to position and cement the place of Costa Rica as a safe biotechnological or medical tourism hub, thereby disregarding ideological debates arising in other countries regarding the restrictions on the use of stem cells. In Turkey, the use of stem cells has been open and regulated as long as they are not embryonic cells, giving way to laws increasingly open to medical tourism [28]. South Korea is a pioneer in the production of stem cell-derived products, [29] notably a composite of allogeneic umbilical cord blood-derived mesenchymal stem cells and hyaluronate to treat cartilage defects in osteoarthritis [24, 30]. In 2014, Japan introduced a “fast-track” regulatory path for regenerative medical products in the Pharmaceutical and Medical Devices Act [31], whereby products can be commercially available following a short trial: patients recruited into that scheme are enrolled in a registry to be followed up for a period during which efficacy must be demonstrated. This law and the subsequent applications of this law were criticized in the international scientific community, [32, 33] using terms such as an “obsession [with staying at the forefront of regenerative medicine]” [34]. Nevertheless, Japan stood its ground [35] and continued research with induced pluripotent stem cells, announcing the start of prospective trials for spinal cord injury and heart conditions in Japanese university hospitals [36, 37], among others. Japanese scientists appear to have followed up on these developments and have published some relevant self-reflection [38, 39, 40].
Irrespective of stem cells, medical tourism is hardly a new phenomenon, and a wealth of literature has been written about ethical considerations and repercussions both in the country of origin and the destination country [41, 42]. Certain countries such as Thailand, Singapore, and India have developed this medical tourism industry to the extent that it represents a non-negligible percentage of their GDP. Contributors to the debate should examine their own biases when referring to foreign countries, avoiding terms such as ‘third world’ or assuming higher possibilities of substandard care, infections, or fraudulence as this may alienate so-called developing nations from much-needed transparency and open cooperation. The perspectives of physicians or researchers from “destination” nations must be included and perhaps should even be the primary source for scientific output, as they should be considered capable of producing critical literature from their own cultural and scientific perspectives. There is a marked inequality in availability and volume of research produced in more affluent countries compared to underprivileged ones, [43] though a process of “catching up” appears to be in place [44].
Nevertheless, medical tourism is a cause of concern, [45] particularly in the case of stem cell therapy, since prospective patients may not be sufficiently informed about the risk and benefits of the treatments they are receiving, or may be charged disproportionate amounts for dubious or unregulated procedures – a form of preying on their state of mind due to their illnesses. Many patients who travel abroad to be treated with stem cells do so as a last resort and in some cases desperately seek a cure or at least an improvement for their medical condition. However, medical malpractice or dishonesty can occur regardless of whether a country approves a given drug or procedure. While medical tourism may contribute or aggravate the proliferation of questionable practices, it can hardly be considered the only reason. Every country has an obligation to implement and uphold good medical practices promoting principles of medical ethics and the deontological standards of medicine. Moreover, the proliferation of fraudulent clinics in some destination clinics is likely hindering the development of legitimate stem cell research [45]. The case of Stamina in Italy is an example where internal regulations successfully identified a fraudulent clinical practice; this operation needed to be halted because of an individual holding no medical qualifications, poor quality standards for manufacturing and no sound scientific rationale, not because of an inherent evil or penchant for fraudulence in stem cell research.
Should medical tourism then be regulated? There is currently no consensus or homogeneity in criteria among countries on regulatory affairs of therapeutic stem cell treatment. The implantation of international regulations would not be a trivial exercise. The suggestion of a recognized agency such as the FDA or the European Medicines Agency (EMA) granting certificates of approval to international clinics, while desirable in theory, would not be possible in practice as it would interfere with the sovereignty of each territory to regulate and legislate their internal affairs. Similarly, the jurisdiction of national agencies is limited to their borders, and allocating a portion of their budget to oversee practices in other countries would not be economically feasible. Some international networking organizations such as the International Society for Cell and Gene Therapy (ISCT), have recently established working groups to tackle regulatory and ethical issues with committee members primarily based in North America, [46] but unlike charters of international intergovernmental organizations, binding for the undersigning nations, their advisories are ultimately non-binding. The Pan American Health Organization (PAHO) has issued recommendations for the use of “advanced therapy medicinal products”, [47] acknowledging that regulatory bodies are still developing in many countries and stressing that ethical implications must be considered, that treatments must take place in authorized, specialized centers, and that a risk-based approach could be considered for establishing regulations. This document notably states that “continual advances in scientific research generally keep it several steps ahead of regulatory mechanisms, a situation that occurs worldwide,” and calls for open communication between scientists and regulatory bodies [47].
Another possibility to curb medical tourism would be to introduce restrictions into the national health network for those patients who have traveled and received treatment in clinics unrecognized by regulatory bodies of e.g., the USA – such punitive measures might, however, not be conducive to the best interests of the patients. Patients may feel they have to lie or be reluctant to request their physician’s opinion, thereby losing an opportunity for education or more extensive research into the treatments they seek abroad. A more constructive solution would then be to actively educate prospective patients before their decision, ensuring that the cost-benefits of stem cell treatment are carefully considered, including the possibility that effectiveness may be small, limited, or non-existent for their particular pathology. The education of patients is hardly a novel idea [48]. The information should be presented in a clear, easily understandable manner rather than a dry scientific report, as this is likely not the format that prospective patients are used to consuming in an era where multimedia is pervasive. Neither should the tone of the information provided be perceived as scolding or belittling, as this may have the unintended effect of the valuable advice being dismissed altogether to the detriment of the patient; efforts should be focused on empowering the patients to make their own decisions (to “know best” or to “be in the know”) – or any other desirable traits that a sociological study into the characteristics of this population may reveal, along with decision-aid studies. For example, they could be more receptive to someone they perceive as a peer than to a disengaged physician or a removed academic seemingly removed from the daily struggles of their conditions. The same narrative and visual techniques employed to attract patients can also be used to educate. Recent efforts, e.g., by the International Society for Stem Cell Research (ISSCR) [49], remain very text-heavy and potentially unengaging for the general public.
However, if after considering the risk/benefits, the patient still decides to seek treatment abroad, be it a clinical trial or a treatment approved by the destination country’s regulatory bodies, at a cost or not, it then becomes the right of a patient to health and the right to access health, to improve their quality of life – this is, perhaps, far more complex to regulate or legislate in an international context. There is a delicate balance between governmental control (imposition or restriction) of therapeutics and the reach of these controls inflicting upon the freedom of the patients to choose what they (along with their medical doctors) want to pursue for their health, under the sole assumption that the patients are fully informed about the risk and possible benefits associated with the treatment.
4. Let doctors be doctors: the patient-doctor relationship
The 2018 Right to Try Act of the USA creates a legal framework for access to unapproved medical products by patients with life-threatening illnesses who have exhausted all their options and may not participate in a clinical trial, provided said product has completed a Phase I study [50, 51]. Opponents of this law have argued that it creates conditions for physicians to prey on desperate patients by creating false hope, that the burden of treatment costs is shifted to patients and manufacturers, and that existing health disparities may be exacerbated, ultimately leading to greater patient suffering [52, 53]. Additionally, valuable information or data collection about product development or adverse events may be lost due to the lack of FDA oversight [52]. The legislation does not compel a manufacturer to provide access to treatment; some manufacturers may outright refuse, [54] and early reports seem to indicate that drugs are still being requested in greater volume under Expanded Access rather than with the Right to Try provision [55, 56]. This legislation raised much controversy, often politically charged, sparking ethical debates about what it was trying to achieve, how much it would truly help, or how it would be implemented in practice. The ethical problems of stem cell therapy then seem no different from that of any other experimental therapy (such as those for cancer or rare diseases contemplated by this law) where the patient’s autonomy, the cost/benefit of the treatment and any possible abuses or misuses must be weighed in. Advocates of the Right to Try Act emphasized the liberty of patients to choose a treatment and to eliminate bureaucracy; in a similar vein, Texas House Bill 810 (85R) authorized the “provision of certain investigational stem cell treatments” under investigation in clinical trials to patients “with certain severe chronic diseases or terminal illnesses.” [57]. The patients were required to provide written informed consent, and the treatment was to be overseen by an Institutional Review Board (IRB), and administered by a certified physician in a hospital, surgical center, or medical school. The IRBs were to submit annual reports of treatments enacted under this law. “Why can’t someone that is of age have the ability to sign off, so to speak, with regard to a proper medical release on the ability to do something that can make such a dramatic difference in their life, and their lifespan, and their quality of life?” argued the proponents [58]. This would also encourage “medical innovation” [58] in the state of Texas – not unlike the intent of countries who have passed similar laws allowing stem cell research or therapy. These two landmark legislations of the United States exemplify a movement to put the patient and their doctor’s relationship at the forefront, which has not been without controversy [59, 60].
If a therapy exists, every effort should be undertaken to implement a way to access said therapy as a last resort for patients. The requirement of having successfully completed Phase I of scientific research may be a reasonable compromise, as long as the patients are sufficiently and objectively educated about the risks and cost/benefits before reaching a decision. And yet, as the example of Japan has shown, a national law, a patient registry, and clinical trials overseen by universities, have still not been considered enough by the academic community. When is it enough? What and whose criteria drive this quantification? Would the debate not be enriched from the participation of physicians and patients who are, so to speak, in the front lines of the battle? One may argue that a “desperate” patient cannot adequately provide informed consent due to their state of mind, but this is a particularly thorny argument that toes close to discrimination, paternalism, [61] or, to use a more modern term, ableism: is having an illness ever sufficient to render a person incapable of making decisions regarding their own welfare? Is a person’s dignity and mental ability lessened or invalidated when faced with a significant loss of quality of life or an eventual end of life? This decision-making process perhaps belongs more in the sphere of a qualified psychiatrist or therapist for each particular case rather than a broad stroke ruling in an academic setting or legislated by a government body. Broadly qualifying a disadvantaged person (in this case, one with an illness) as intrinsically “vulnerable” or incapable of making informed decisions for themselves may be an insult that reinforces the injustices and stigma that they already face [62]. A decision to use or seek new treatments is not in itself irrational; if prospective patients would be considered competent enough to consent to enter a phase I/II trial in a research setting, why could they not consent in a non-research setting under a physician’s supervision? [63]. And if patients have the right to refuse a treatment, surely from this right to refuse follows a reciprocal right to choose or access an intervention [59, 64, 65]. It is an infringement on the right of patient to procure treatment that they understand would be useful for their condition after being presented with clear and truthful information about said treatment. Instead of paternalistic protection, prospective patients need empowerment: a greater voice in setting research agendas and designing studies [62].
Perhaps the most common concern is the potential harm of stem cells themselves or the problems of their commercialization, as the non-maleficence principle must always be kept in mind. The relative safety of mesenchymal stem cells has been sufficiently covered, as reported by systematic reviews [21, 22, 23]. Still, more research is needed for standardization of dosage, culture methods, and source of the stem cells, as well as a need for quantifying effectiveness for clearly defined conditions and thorough documenting of adverse events. Regarding commercialization, as long as there is a demand, there will always be a market to fulfill that demand with various degrees of ethical and legal shades. Stem cell therapy is no stranger to such a conundrum in the face of market greed. A problem of commercialization may derive from health providers being unwilling to fully inform patients about the risk/benefits for fear of losing business; however, not all practices are the same, comparable to medical practices running legally in which their marketing strategies might present skewed information to capture more clients. Within legal boundaries, there could still be unethical behaviors and vice-versa. On the other hand, patients who have gone to great lengths to receive treatment may experience a placebo effect or convince themselves it was worthwhile. Yet incurring in costs to access a treatment is not inherently unethical or fraudulent; the FDA has published a guidance outlining requirements where this practice may be authorized, notably when the costs would be extraordinary to the sponsor because of “manufacturing complexity, scarcity of a natural resource, the large quantity of the drug needed (e.g., based on the size or duration of the trial), or some combination of these or other extraordinary circumstances (e.g., resources available to a sponsor)” [66].
The pressure to find new therapies for illnesses with limited, insufficient, or no current treatment options comes precisely from the physicians, the scientific and medical industry, and patients seeking relief for their conditions. Herein lies the more significant risk: a race for supply and greed of demand when faced with pain or eventual death. Suppose a particular country’s laws or guidelines are restrictive enough to hinder the physician/patient relationship. In that case, doctors may find it impossible to consider alternative therapies, even under compassionate use, due to the lack of adequate protocols. At the same time, patients feel powerless in the face of government regulations. It is precisely at this point where, in desperation, abuses or misuses arise, not from the new therapies themselves, but from a lack of expectations or incomplete information about the possibility of a cure or relief. And one may ask: what’s the rush? Why do some patients insist on seeking a treatment that is not readily available or approved instead of waiting for the due process of clinical research? “The rush is the daily necessity to help sick people. (…) The ‘rush’ arises from our human compassion for our fellow man who needs immediate help,” as “their illnesses will not wait for a more convenient time” [67].
If a legally qualified doctor in his professional authority, after having read results of recent advances in the field, well within the boundaries of the regulatory bodies of the country where they operate concludes that such treatment could help a particular patient, a patient who is willing and fully informed to the best of the current understanding – would they be morally justified in refusing said treatment? Can moral objections ever be a sufficient basis for denying the right to healthcare? Patients who seek stem cell treatments may have spiritual distress or therapeutic hope, [68] an aspect some medical doctors may not be equipped to manage. But what of compassion? This use of “compassionate” may be close to that employed by ecclesiastical authorities who do not oppose but promote treatments that offer at least a better quality of life: a physician’s duty is not limited to knowledge and technical expertise, but also compassion [69]. The compassionate act and the treatment as compassion, coming from the good judgment of a doctor seeking the best for his patient, must therefore be left in that sphere of the doctor’s relationship with his patient. Depersonalized rulings do not allow the physician to exercise his art, profession, and oath. The relationship between the physician, or a team of physicians, and their patients is thus an essential aspect of making an informed decision: one the one hand, the patient exercising his autonomy to make medical decisions, and on the other, the physician, upholding his medical oath and not creating false expectations by promising more than what is expected to be achieved with a given treatment or to create hopes beyond what can be offered. Thus, the final decision to access an intervention must lie in the hands of the patient and their physician, based on real world evidence for safety, and within a sound legal and ethical framework.
“Ethics in both research and clinical settings is most effective when it is preventive” [70]: indeed, bioethicists do not go ahead with scientific developments but discuss scientific issues that are already on the table. Conversely, neither should physicians regard ethical questions as “removed from their daily work at bench or bedside,” as the purpose of new treatments is a societal benefit [70]. “Market will efficiently allocate the resources, but not always in an ethical manner” concerning medical tourism; [41] ethical considerations should therefore be contemplated before the application of treatments by creating a legal framework that promotes scientific research and keeps the welfare of the patient at the forefront. The fear of possible misuse or medical misconduct should not deny the patients’ right to health, particularly when their lives are at risk or when they are the most vulnerable to their condition. Accumulated clinical experience and evidence-based medicine about safety, dosage, and efficacy would be more appropriate when determining whether to offer or withhold access to treatment.
5. Legal and moral issues
The international legal system is derived from ethical principles, at the heart of which lies the dignity of the human person, and modern bio-law too draws from ethical and axiological foundations. Human dignity as a concept is fundamentally imprecise, as its definition necessitates defining first what is dignified or worthy, and what is unworthy – an anthropocentric, Judeo-Christian notion [71]. Subsequently, the western concept of human dignity has evolved and dissociated itself from any deity to accept the teleological interpretation of the en