Your safety is our commitment.
At Riordan Centers for Cellular Medicine, safety isn’t just a requirement, it’s our promise. From the moment cells are sourced to the moment they’re administered, every process meets the highest scientific, ethical, and regulatory standards. Our clinical and laboratory teams work seamlessly together to ensure every treatment is tested, traceable, and delivered with uncompromising care.

Certified excellence
Our lab operates under GMP-compliant and ISO-certified systems and follows strict documentation, validation, and quality control processes to ensure every cell is pure, potent, and consistent.

Trusted clinical care
Safety extends beyond the lab. Our physicians and clinical specialists uphold the same rigor in patient care, following evidence-based protocols for preparation, dosing, and monitoring. Every treatment is guided by experience, compassion, and a commitment to the highest standards of safety and results.
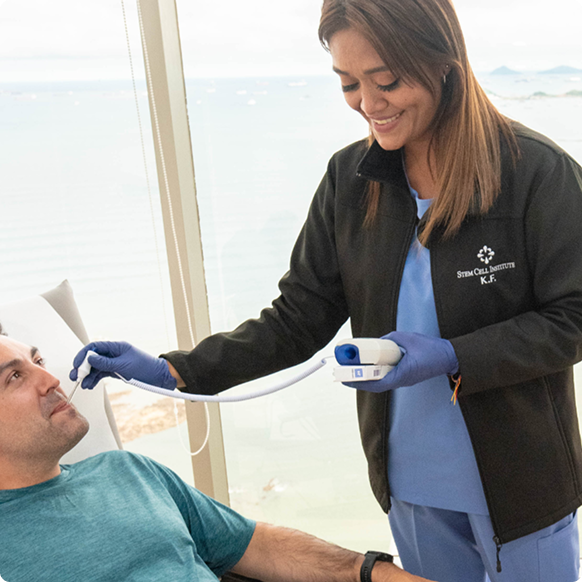
Tested & proven
Before reaching a patient, every cell undergoes comprehensive safety, sterility, and potency testing. Each step is verified for identity, purity, and viability—ensuring every therapy meets our clinical and scientific benchmarks for quality and effectiveness.

Ethical oversight
Integrity guides everything we do. Our cell sourcing, processing, and clinical protocols are built on ethical transparency and are reviewed by independent oversight boards. From donor to patient, every step is designed to protect and respect human life.